What is Phenol Red?
Phenol Red is a water soluble, color changing dye. It is most commonly used as an indicator. It is found in a red crystal form and it is a weak acid. It is also known as phenolsulfonphthalein or PSP.
What is Phenol Red Used For?
Phenol Red is a general purpose pH indicator generally used in cell biology laboratories. It can be used well because it is a water soluble dye. Phenol Red turns yellow when it detects a pH of 6.8 and it turns red when it detects a pH of 7.4. Also, when the pH reaches 8.1, it turns a bright pink color. Because of the difference in colors, it is an effective pH indicator. It is commonly used in pool test kits as well.
Invention of Phenol Red
Phenol Red was first made in a lab. I conducted research but could not find anywhere who it was first made by, when it was first made, or where it was made. It is not a very well known molecule which is why this probably isn't known. It has been more popular recently since the dangers of it have been discovered and they realized how to prevent them.
Formation in Nature
Phenol Red is not found in nature and can only be made in laboratories.
Chemical and Structural Formulas
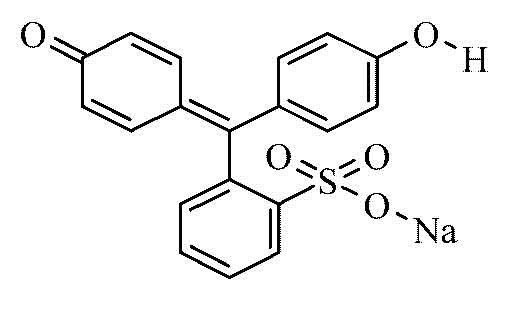
Phenol Red's chemical formula is C19H14O5S.
Phenol Red's structural formula is:
Phenol Red's structural formula is:
Exposure or Disposal Problems
Phenol Red is both corrosive and toxic. This means that if it touches the skin it can burn through it and if swallowed it can cause problems as well. Deaths have been caused if it is on more than 25% of the skin area or if as little as 15 mL are consumed. If safety glasses/goggles and aprons are worn, this material isn't very dangerous. These dangers are the reason why it is mostly used in labs.
Reactions that Create Phenol Red
The reactions that create Phenol Red are when all of the elements within the molecule are combined in a laboratory with the help of technology. There is no natural way of making this, it is an artificial molecule.